Galvanic corrosion
- Title: Galvanic corrosion
- Subtitle: Melisa
- Content text:
Galvanic corrosion
The human body is not a hospitable environment for an implanted metal alloy: a highly oxygenated saline electrolyte with a pH of around 7.4 and a temperature of 98.6°F (37°C). All devices will be subject to corrosion inside the body, releasing metal ions, which may interfere with physiological functions. In 2019, the US FDA said the following about how metal corrosion leading to an increase in metal ions can affect human health: “When released in sufficient quantities, these corrosion by-products may lead to adverse biological effects.” (1)
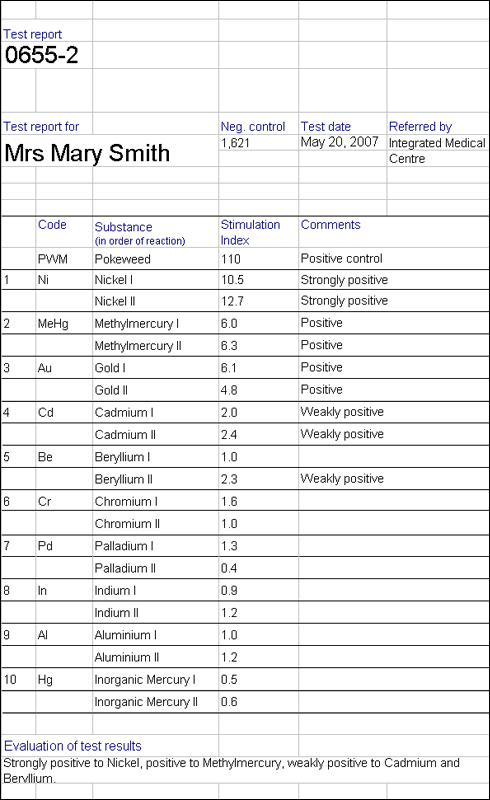
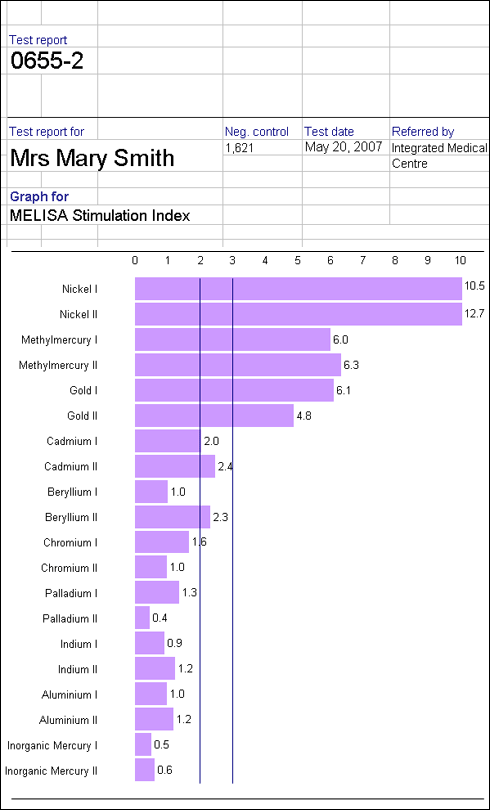
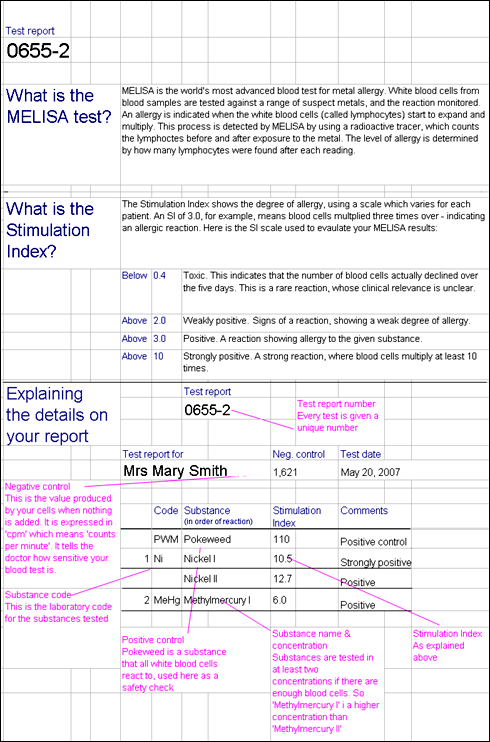
It is important to consider total exposure to metals when testing for hypersensitivity. Although symptoms may appear when, for example a titanium implant is placed, it is possible that the reaction is more complex, and that the new implant may contribute to a hypersensitivity reaction to metals previously tolerated at lower levels. Certain combinations of dissimilar metal alloys, may lead to increased corrosion. For instance with a combination of stainless steel and cobalt chromium or titanium alloy, the stainless steel will corrode more quickly. As stainless steel contains 10% nickel this may be problematic for these with nickel hypersensitivity.
Galvanic corrosion in dentistry
In dentistry, a galvanic current is created when there are dissimilar metals in the mouth, for instance gold crowns and amalgam fillings. The effect is like a battery in the mouth, with saliva acting as an electrolyte. Thus, the corrosion of the alloys – and subsequent release of metal ions – is increased.
Toothpastes and mouthwashes containing fluoride can also increase corrosion in the mouth (2,3,4). Eating hot and cold food, changes in pH (alkaline products like milk or acidic like tomatoes and dressings) all speed up the corrosion process (5).
You can read more about galvanic corrosion and oral galvanism:
- The International Academy of Oral Medicine and Toxicology (IAOMT) website: https://tinyurl.com/4h8765az
- Watch a video about biocompatibility & oral galvanism: https://tinyurl.com/6k2266ex
- Watch a video on sensitivity to dental materiale: https://tinyurl.com/yxhh76xe
Systemic galvanic reactions
Dr Scott Schroeder, a foot and ankle surgeon, has been involved in a 12-year study which has included the surgical removal of over 1,000 implants in over 400 patients.
The research was initiated when Dr Schroeder was following up with his patients and found that a surprisingly high number had significant life-changing negative effects post-operatively, after foot and ankle surgery involving metal hardware. Wondering about the causes, he started to look more widely into the issue and investigated what other types of metals they had in their bodies, including in their mouths.
In some cases, he observed “what certainly appeared to be galvanic reactions between dissimilar metals in different parts of the body which were NOT in contact with each other. In these cases, metal I removed from the foot had incredibly significant positive life changing effects, not in the foot but in other areas of the body where symptoms were present.”
You can see Dr Schroeder’s testimony to the FDA about the adverse effects of metal implants and how they may contribute to fibromyalgia, migraines, nerve symptoms, joint pain, severe fatigue, dermatological issues, exacerbation of rheumatic conditions and joint pain in susceptible patients here: https://www.youtube.com/watch?v=iYa5nb1xjvg
References
[1] FDA Biological Responses to Metal Implants. 2019
[2] J. Noguti, F. de Oliveira, R. C. Peres, A. C. M. Renno, D. A. Ribeiro, The role of fluoride on the process of titanium corrosion in oral cavity, Biometals 25 (2012) 859-862.